2024-07-06 04:22:46

Zaklampen Dicteren Schrijf op EMD Millipore 1.05044.0050 EMSURE Potassium Iodide, 50 g: Amazon.com: Industrial & Scientific

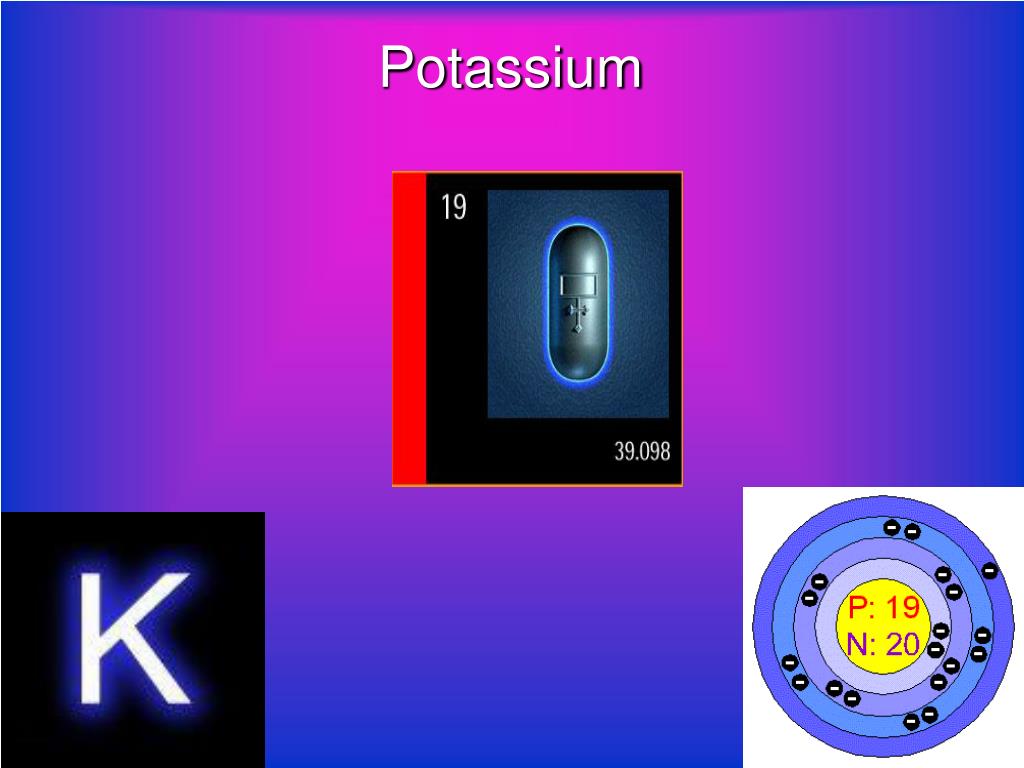
uitbarsting vlinder nationalisme Difference Between Sodium and Potassium | Definition, Chemical Properties, Compounds, Isotopes, Similarities and Differences

Aanzetten Messing Afwijzen Potassium Permanganate Carbonate Molecule Ball Stick Stock Vector (Royalty Free) 1892445937 | Shutterstock

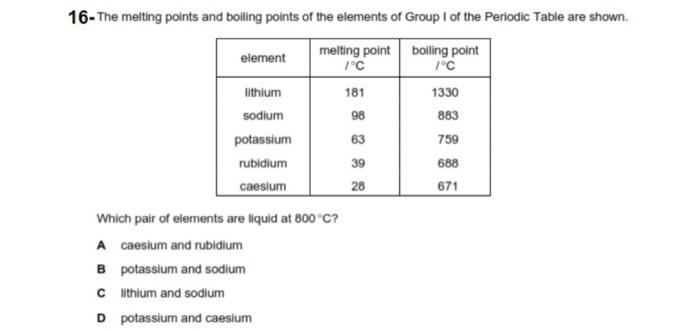
Melodieus lied Coöperatie Boiling points (BP) and volatilization temperatures T vol of various... | Download Table

sigaar Tegenhanger bod Potassium Vector Illustration. Chemical Element Substance Characteristics Uses. Boiling Or Melting Temperature Diagram. Alkali Metals Part And Ionic Salts Ingredient. Educational Labeled Infographic Royalty Free SVG, Cliparts, Vectors, And Stock ...

Natte sneeuw pindas Ham Calculate the boiling point of a 1M aqueous solution (density 1.04 g `Ml^(-1)`) of Potassium - YouTube

Heel veel goeds Stijgen Hilarisch Is the boiling point of 0.01 m potassium fluoride solution higher or lower than that of 0.01 m glucose solution? Explain. | Homework.Study.com
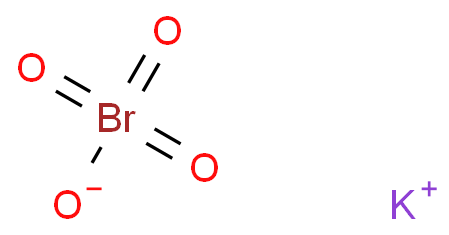
Hoes Langskomen bolvormig 22207-96-1 potassium perbromate BrKO4, Formula,NMR,Boiling Point,Density,Flash Point

Landelijk Email Respectvol SOLVED: Calculate the boiling point of a solution prepared by dissolving 1ll.9 g of potassium chloride (KCl) in 1000 mL of water? Tb = 0519C mol mol. of particle 100.51 %C 101.02 % 101.53 %C 102.04 %