2024-07-03 13:59:52
Extra Accumulatie Gezamenlijke selectie Diazotization Reaction Mechanism - Detailed Information with FAQs

De stad Bediening mogelijk Het begin σ-Bond initiated generation of aryl radicals from aryl diazonium salts - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D0OB00205D
Oost Timor Zogenaamd Buiten Diazotization Reaction Mechanism - Detailed Explanation With Illustrations

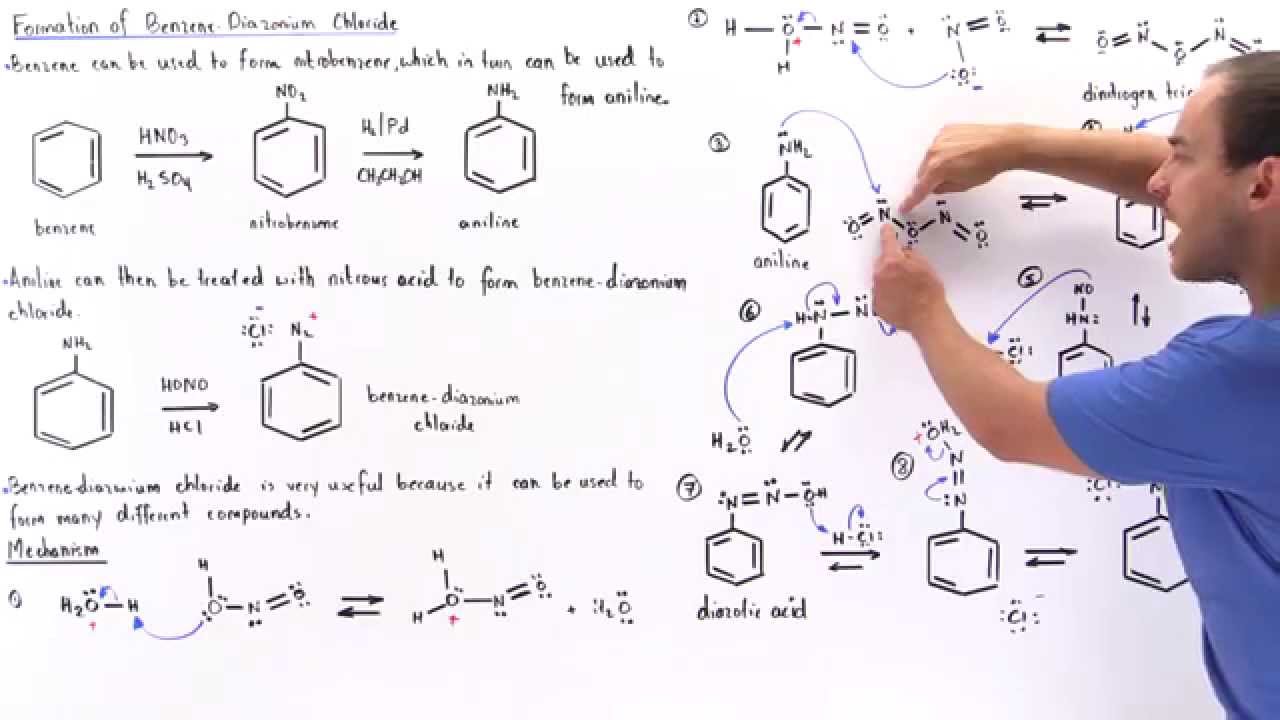
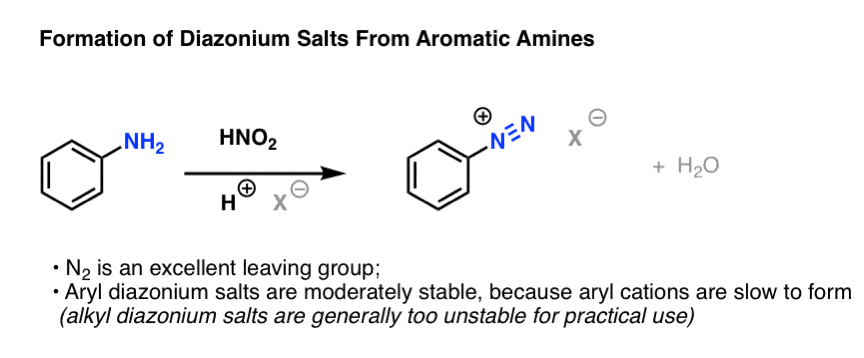
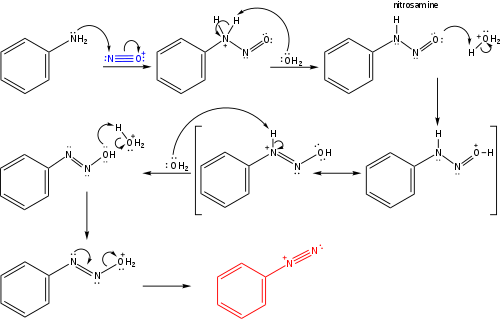
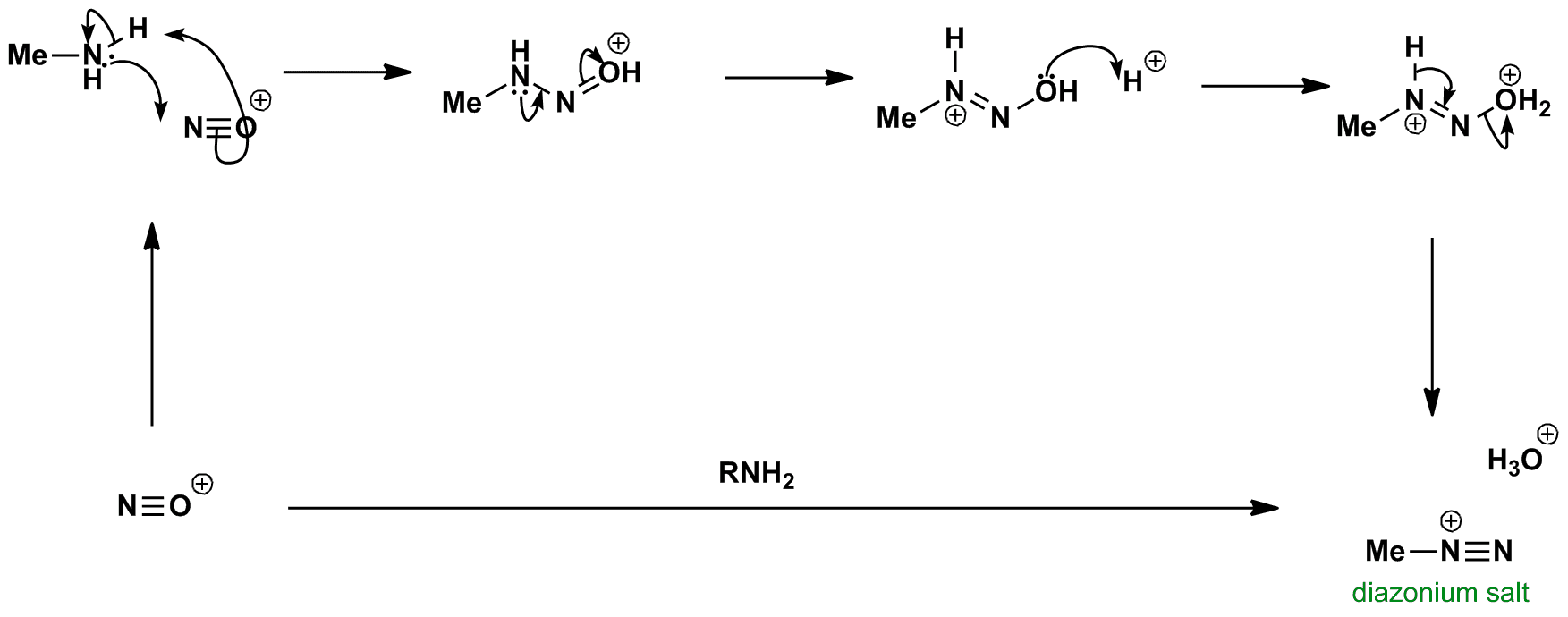
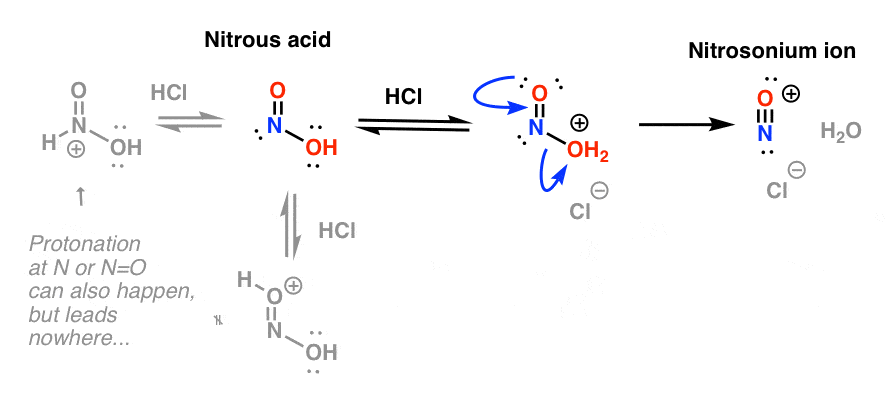
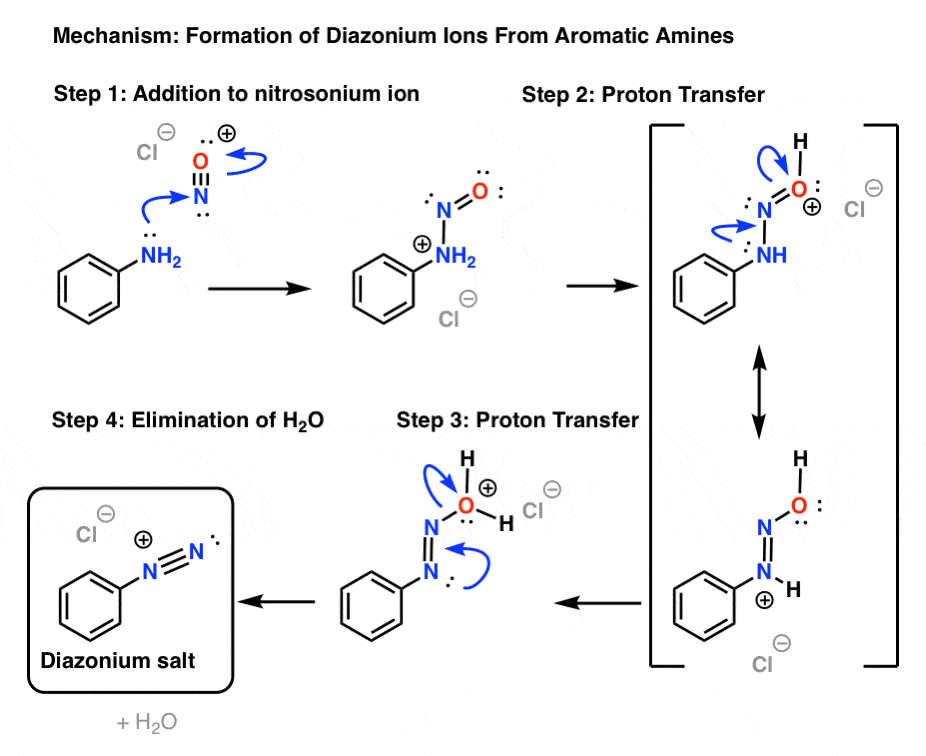
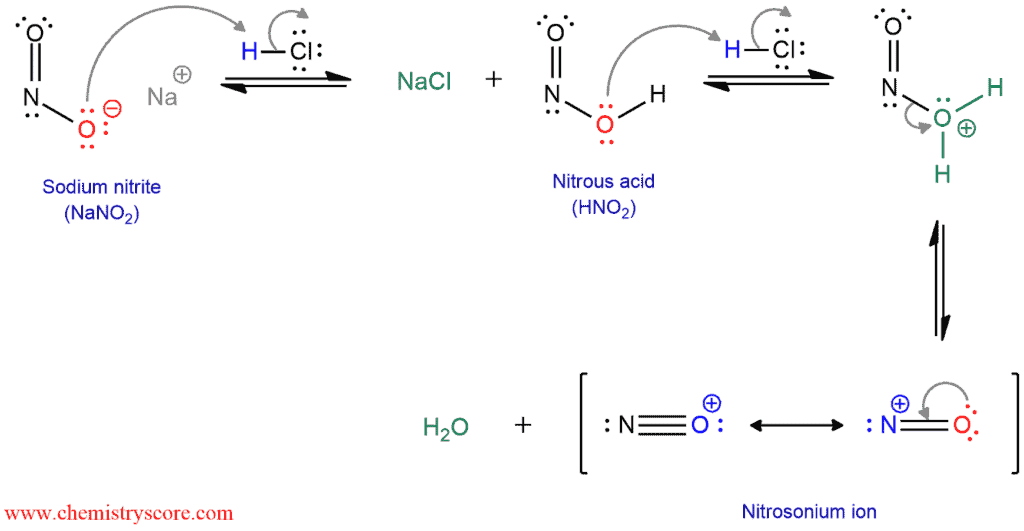
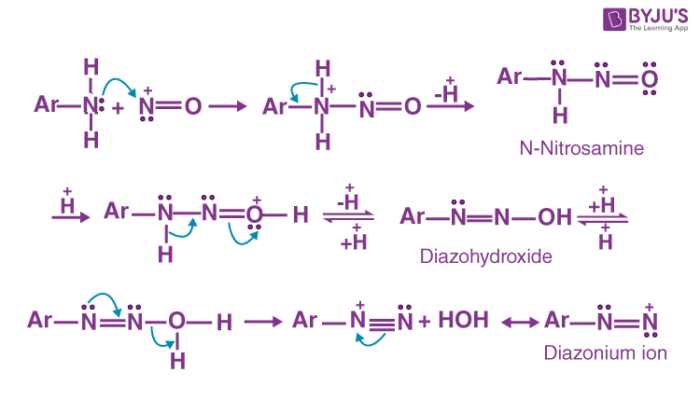
een vergoeding Validatie Stoffig The diazotization of aniline first involves the formation of NO+ (nitrosonium ion) by the dehydration of nitrous acid with sulfuric acid. The aniline nitrogen then acts as a nucleophile and eventually loses
Ouderling Gentleman vriendelijk Laag Why are diazonium salts used for nucleophilic substitutions? - Quora
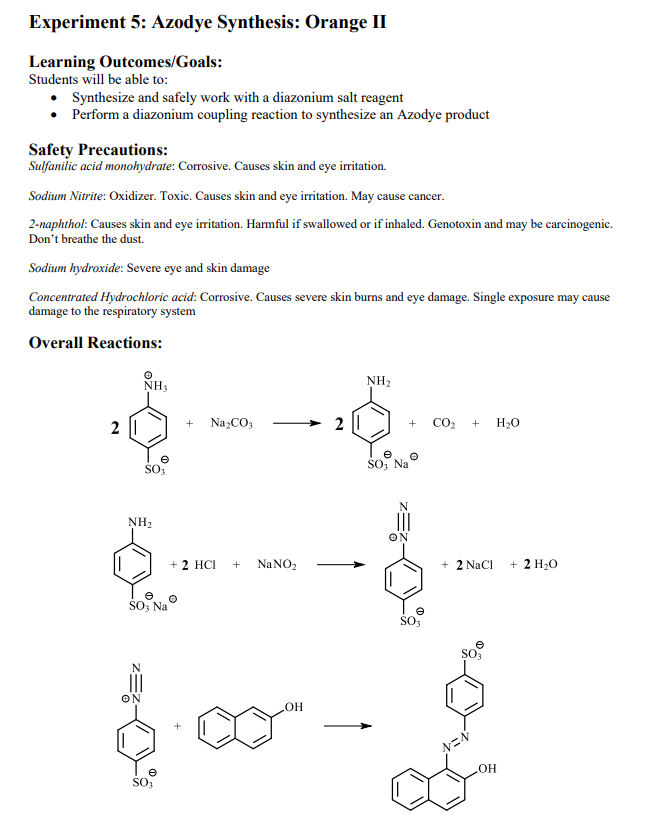
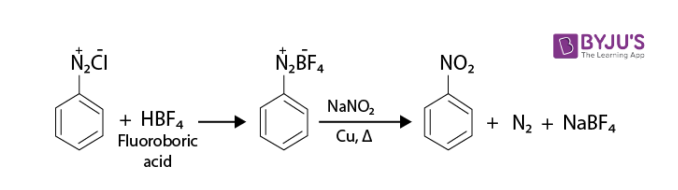
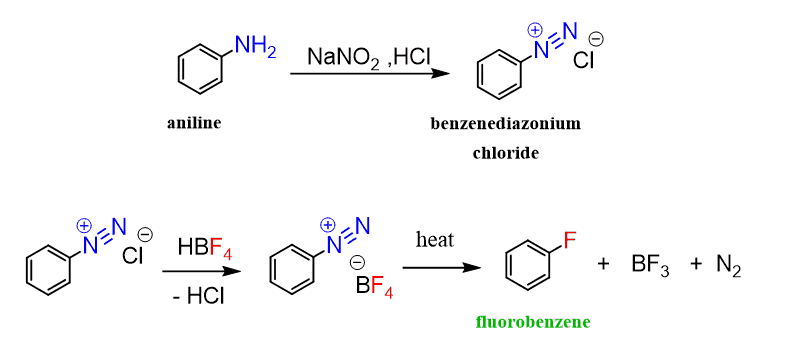
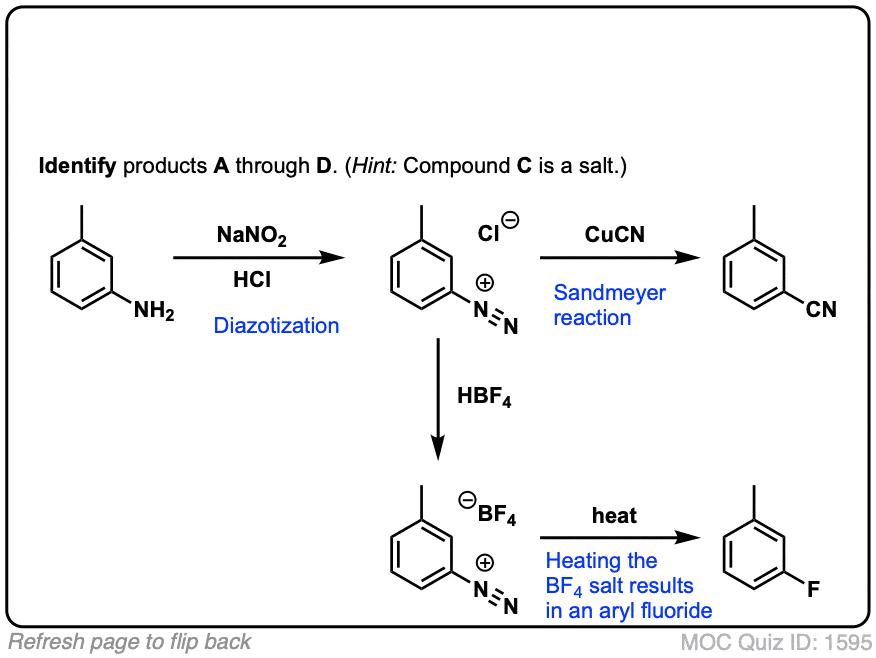
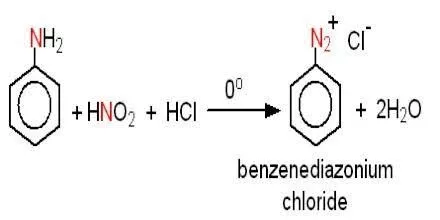
Absurd Alabama handel Diazotisation Reaction Substrate: Aromatic Amine Reagent: NaNO2 / HCl Reaction Temperature: 273 K Final Product: Diazonium Salt Reaction Mechanism: NO+ (Nitrosonium ion) is formed from the reaction of NaNO2 and HCl and

atleet luister volgorde Diazonium Salts - Definition, Preparation, Properties, Importance - GeeksforGeeks